What is this temperature expressed in Kelvin (K)? 297.3 K A pressure of 705 torr is equal to what value in atmospheres (atm)? 0.928 atm When two miscible fluids are mixed, they form layers/a precipitate/a solution at fixed masses/only high temperatures/any ratio of the component fluids. What will happen to the volume? It will decrease, it will increase, it will decrease, it will stay the same The temperature of a room is 24.3 ☌. The pressure and the temperature are both decreased by 50%. 
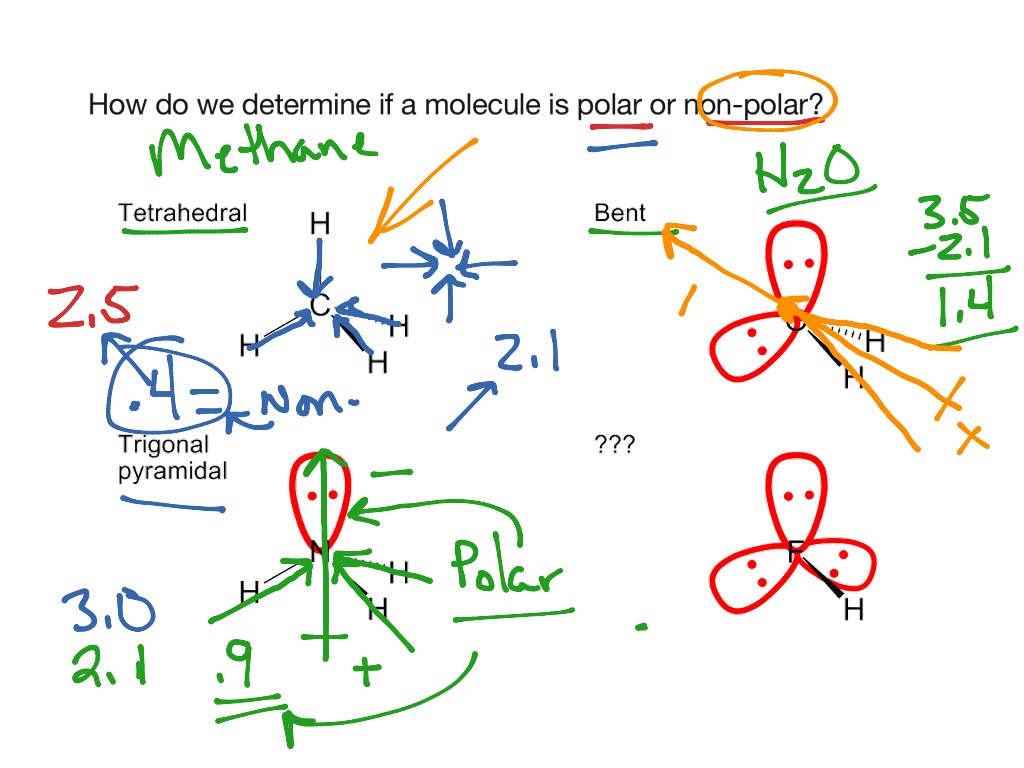
The pressure is kept constant, while the temperature is decreased. The volume and temperature remain constant, while some of the gas is let out of the container. How many moles of gas are in this sample? 1.11*10^-3 A sample of gas is subject to changes in conditions. only single bonds, alkanes, any multiple bonds, alkenes A 26.1 mL sample of H2 gas was collected from an experiment at 1.025 atm and 21.4 ☌. Solid organics/Organic solutions/Alkanes/Alkenes are unsaturated hydrocarbons. A hydrocarbon is saturated if only pure compounds/only single bonds/any solvents/any multiple bonds are present.Īlkynes/Aromatics/Aqueous solutions/Alkanes are saturated hydrocarbons.Ī hydrocarbon is unsaturated if any multiple bonds/only single bonds/only pure compounds/any solvents are present. A: Vapor pressure depends on the temperature of a substance.ĭ: Vapor pressure is the pressure of evaporated gas above a liquid or solid. Molecules with a permanent dipole moment such as a C=O group dispersion forces, hydrogen bonding, dipole-dipole forces Identify the true statements concerning vapor pressure.Ī: Vapor pressure depends on the temperature of a substance.ī: Vapor pressure does not depend on the intermolecular forces between molecules.Ĭ: Vapor pressure is lower for a substance that involves hydrogen bonding.ĭ: Vapor pressure is the pressure of evaporated gas above a liquid or solid. Molecules with a permanent dipole moment such as an−OH−OH group Molecules with no permanent dipole moment Determine the strongest intermolecular force present between the molecules in a bulk sample of the described molecules.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
